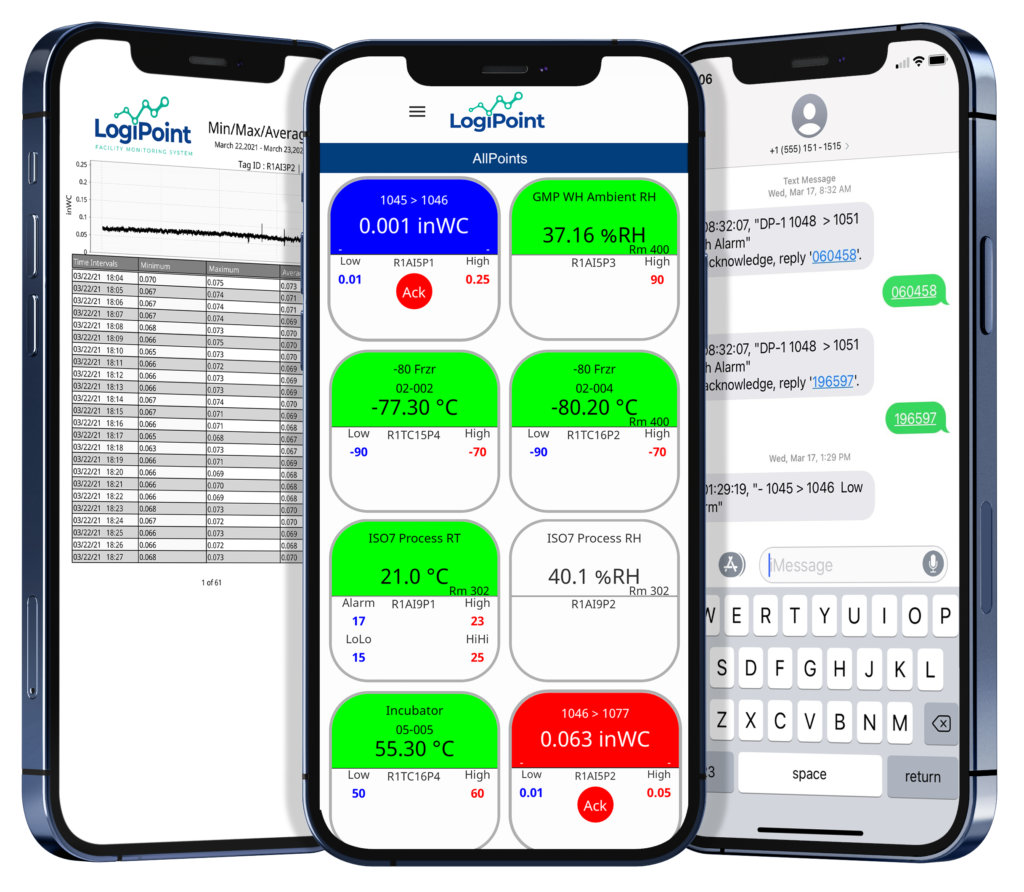
With LogiPoint®, you get more than just numbers on a screen:
- Audit-ready reports at your fingertips
- 24/7 environmental monitoring across all critical systems
- Redundant safeguards that protect against network or power loss
- Scalable coverage, from a single freezer to multi-site facilities